Health & Nutrition
Antinutrients
Every food in the world comes with natural negatives attached to it, in the form of chemical compounds that are bad for us in some way. These natural negatives come in a variety of types including antinutrients, toxins, and carcinogens. As the name suggests, antinutrients are compounds that interfere with the absorption of nutrients. Different types of antinutrients act in different ways, but the common forms of antinutrient action include inhibiting the action of digestive enzymes, interfering with the absorption of minerals, and even making plants taste bad so we don't want to eat them. This article will deal with the antinutrients that get the most attention in the pet bird community.
Oxalate
Oxalic acid is a compound found in many plants that has a strong affinity for binding with minerals. Its calcium-binding properties get the most attention because calcium oxalate is the primary ingredient in human kidney stones, and calcium is the mineral of greatest concern in the diet of pet birds. Oxalates "tie up" the minerals that they bind with; the body can't absorb these oxalate-mineral compounds so they are passed out of the body without being used.
In the pet bird community there's a great deal of concern about oxalates causing calcium deficiency in birds, but this concern seems to be misplaced. In the human health community there doesn't seem to be any concern at all about calcium deficiency; instead the focus is on the role that oxalate plays in the formation of kidney stones and other health problems including gout. But apparently oxalate-related kidney stones are not an issue with birds. Kidney stones and gout in birds are called urolithiasis, and it's caused by excessive uric acid not by oxalate consumption. I can find no evidence that birds get calcium oxalate kidney stones as humans do. Bird kidneys function in a way that is fundamentally different from mammals, so it's not surprising that the way they process substances like oxalate may be different from humans.
Bird owners tend to be concerned about the amount of oxalic acid in vegetables, and some vegetables do have a high level of it. Oxalicacidinfo.com has a chart showing the levels in vegetables as measured by three different sources, and the different sources came up with radically different amounts for some vegetables. The site notes that the these numbers may not be particularly meaningful anyway: the type of oxalic acid (soluble or insoluble) in the food has an effect on absorption, growing conditions and cooking method affect the amount of oxalate in the food, and the National Institute for Health believes that oxalate interactions have little or no nutritional consequence for people who eat a variety of foods.
Do you know what else has a lot of oxalic acid? Seeds, whole grains, nuts, and beans (Chai & Liebman, Denver Nephrology, Kaiser Permanente). The bird community is generally unaware of this, even though these are the foods that make up the bulk of our birds' diet in one way or another. Oxalic acid is pretty pervasive in plants and there's no getting away from it. It's highly probable that the plants that make up our birds' wild diet have a similar oxalate profile to what we're feeding them. So birds must have some kind of adaptive mechanism to help them deal with it, and I think I know what it is. It's common for wild birds to eat dirt and grit, which can be excellent sources of minerals.
It would be more helpful to think of the relevant foods as being low in calcium rather than high in oxalate, because that's what the real issue is. Many low-oxalate foods are also low in calcium, but we don't freak out about those. The most obvious solution is to keep an eye on the calcium content of the overall diet to make sure that your bird gets enough. Giving your bird access to free-choice calcium sources like cuttlebone, mineral block, or mineral grit will go a long way toward making sure they can get enough calcium/minerals to offset the low-calcium foods, as long as they actually use these resources. It doesn't do any harm to try to limit high-oxalate foods, but oxalate is too widespread and there are too many variables that affect oxalate level for this tactic to be very practical or effective.
If I understand the information at Oxalicacidinfo.com correctly, the numbers issued by the USDA and other sources refer to the amount of oxalic acid in a food (the name of the compound BEFORE it has bound itself to a mineral), and not the amount of oxalate in the food (the name that is used AFTER the compound has bound itself to a mineral). However, many sources use the word oxalate for either form, and the rest of this article won't be too fussy about using correct terminology. Side note: there can be quite a lot of pre-existing calcium oxalate (up to 80% of the dry matter) in some plants according to Ruiz-Agudo et al.
I can't find a source that says so directly, but presumably the amount of minerals that the USDA lists as being present in a food refers to the amount of mineral that is not insolubly bound to something at the time the food is eaten. It would be pointless to list mineral compounds that are completely indigestible But once the food is eaten, the digestion process gives the minerals and oxalic acid the opportunity to get together and form a compound.
Sources including the Cleveland Clinic and Johns Hopkins say it's a good thing when excess oxalic acid binds with calcium in the intestines, because unbound oxalate can be absorbed into the body and cause kidney stones. They urge you to eat plenty of calcium, not to prevent calcium deficiency, but because they WANT it to bind with oxalate and be passed out of the body.
Consuming very large quantities of oxalates can cause oxalate poisoning, resulting in kidney damage or death. Rhubarb leaf is considered to be toxic because of the high levels of oxalic acid it contains. There are other plants that contain dangerous levels of oxalate, and even excessive consumption of black tea can cause oxalate poisoning. But these are extreme cases, and oxalate poisoning isn't a concern with ordinary consumption of plants that are generally recognized as food.
It's difficult to figure out how much of the calcium and other minerals in the diet are bound by oxalate. The situation is complicated by the fact that the body manufactures its own oxalate (Holmes et al), and it's believed that 80-90% of the calcium oxalate in kidney stones came from internally-produced oxalate not from the diet (BioIndividual Nutrition).
Chemistry can give us an answer about how much calcium can potentially be bound by the oxalate in foods. Unfortunately my chemistry knowledge is pretty weak, but it goes something like this. Calcium has a molecular (mole) weight of 40 (PubChem), while calcium oxalate has a mole weight of 128 (PubChem), indicating that the oxalate has a weight of 88. So oxalate will bind calcium in a ratio of 8.8 to 4, measuring by weight. Another way of saying this is that 2.2 milligrams of oxalate can bind 1 milligram of calcium.
This is the worst-case scenario as far as calcium is concerned, but the actual amount that's bound by oxalate will probably be less than that. Oxalate binds with mineral ions that have two positive charges (indicated by two plus signs after the mineral's symbol, for example Ca++). Calcium, iron, magnesium, manganese and zinc all occur in this form in the body (IASPOINT) and apparently in plants too. So oxalate can bind with all of these minerals, in the body and in plant foods. I've been told that the oxalaic acid should prefer some ions over others, but I haven't been able to find any information on what its preferences are (Hypography). Riley et al seem to suggest that oxalate might prefer magnesium over calcium, at least during the formation of kidney stones, but I've been told that the paper is ambiguous. They also indicate that other substances (like citrate) can compete against oxalate to bind the calcium. Calcium citrate is highly digestible, so offering a little citrus fruit along with high-oxalate foods might help reduce the amount of calcium that is lost to the oxalic acid.
In any case, calcium ions are much more abundant in the body than any of the others (RSC, LiveScience), and calcium also tends to be more abundant than the other relevant minerals in plant foods. So a large percentage of the oxalic acid will probably end up binding to calcium regardless of what its preferences are, just because there is so much of it available to bind with.
Apparently there are other complicating factors as well. Spinach contains more than enough oxalate to bind ALL the relevant minerals in the plant (NutritionData for mineral data, OxalicAcidInfo for oxalate data). But it's widely reported that 5% of the calcium in spinach is bioavailable, indicating that some of it escapes being bound by the oxalate. The amount of minerals that oxalate theoretically COULD bind is apparently not the amount that it actually DOES bind. There are numerous other factors that influence the bioavailability of minerals (Eufic).
It's difficult to find comprehensive information on the bioavailability of minerals, but charts at Scientific Curiosity, HealthKismet, and Oxford have information on the amount of bioavailable calcium in a number of foods that are relevant to birdkeepers. Wake Health and OxalicAcidInfo have lists of the oxalate content in a number of foods. Oxalate.org has a searchable database showing the oxalate content of more than 750 foods.
Soaking, sprouting and cooking all help to reduce oxalate in foods. Table
2 of
Savage et al shows the effect of cooking on oxalate levels in several
common vegetables.
But as mentioned earlier, you probably don't really need to worry about this
stuff unless you have an issue with kidney stones. The real solution
to the oxalate issue is to not go crazy with high-oxalate foods, and make sure that your bird's intake of calcium and
other minerals is adequate.
Phytate and the Ca:P ratio
Phytic acid is another compound found in plants with a strong affinity for binding with calcium and other minerals, making them unavailable for use by the body. It is a compound made up of phosphorus and inositol that forms in plants (Wikipedia). Phytate is the proper name for phytic acid that has bound itself to a mineral, but many sources don't make the distinction between the different forms and neither will I.
Phytic acid is considered to be a less potent inhibitor of mineral absorption than oxalic acid (Oregon State). Phytate is the primary storage form of phosphorus in plants. Phosphorus is an essential nutrient, but in phytate form it is not bioavailable to most animals (including birds and humans). The digestive enzyme phytase is required to digest phytate, and ruminant animals (like cows) are pretty much the only animal that have this enzyme. Phytic acid has many functions within the plant. It has functions in animal cells too, but the animals have to synthesize it for themselves since they can't absorb it from the diet (Wikipedia).
There are numerous factors that affect the binding of phytic acid to other minerals in the body, including pH, gut flora, and the presence of phenols and tannins. The way it binds with minerals varies depending on which mineral it is, and the process is more complex in some cases than in others. From the human health standpoint, its affinity for binding with zinc and iron may cause more problems than binding with calcium, depending on the relative abundance of these minerals in the diet (Kumar et al, Wikipedia). But most people consume enough minerals that phytate isn't a problem. The phytic acid consumed during a meal may affect the absorption of minerals from that meal, but it doesn't affect mineral absorption from subsequent meals. Phytic acid is beneficial in some ways, to the point that it can be bought in supplement form from vitamin sellers (Precision Nutrition, Authority Nutrition).
The mineral-binding preferences of phytic acid have been identified, and calcium is not its favorite mineral. Bohn et al report that one study found that its favorite was copper, followed by zinc, nickel, cobalt, manganese, iron, and calcium, and another study identified the order of preference as zinc, copper, cobalt, manganese, and calcium. But regardless of these preferences, phytic acid is stored in the tissues to a much larger extent with magnesium, calcium, and potassium.
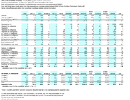 Grains, seeds, nuts, beans and legumes contain far more phytic acid than
vegetables do, and their Ca:P ratio skews heavily toward the phosphorus (see
tables at Precision
Nutrition). The chart at left shows the Ca:P ratio and other nutritional
information for some seeds, grains, and nuts that are commonly fed to pet birds,
with a couple of vegetables thrown in for contrast.
Grains, seeds, nuts, beans and legumes contain far more phytic acid than
vegetables do, and their Ca:P ratio skews heavily toward the phosphorus (see
tables at Precision
Nutrition). The chart at left shows the Ca:P ratio and other nutritional
information for some seeds, grains, and nuts that are commonly fed to pet birds,
with a couple of vegetables thrown in for contrast.
Soaking, sprouting and cooking all help to reduce phytic acid in foods. As with oxalate, providing free-choice minerals will let your bird regulate its mineral intake to offset the mineral-binding effects of phytic acid.
For what it's worth, it looks like the amount of free, non-phytate phosphorus in parrot pellets is about 65-75% of the total phosphorus (chart in the Pellet article). They may have done something special to achieve this level, because Table 2 of a Purdue University article shows that the reverse is normally true - the amount of phytate phosphorus in grains and beans is about 65-75% of the total phosphorus. However the article also indicates that various factors influence the bioavailability of phytate phosphorus, and poultry may be able to use up to 50% of it.
Here are some studies on the effect that phytic acid has in poultry: Adedokun & Adeola, Angel, Onyango et al, Likuski & Forbes, Cowieson et al.
Ca:P. It is often recommended to maintain a calcium to phosphorus (Ca:P) ratio in the diet of about 2.5:1, but it is not clear whether this has anything to do with phytic acid, or whether the phosphorus amounts listed in the USDA food database even include the phosphorus in phytic acid. The recommended Ca:P ratio is similar to the Ca:P ratio in bone (Bonjour), which may be a lot more relevant. Calcium and phosphorus are both essential nutrients that have a complex chemical relationship, and too much of one will disrupt the body's ability to use the other.
In any case it appears that the Ca:P ratio is most relevant during bone growth in infants and juveniles, and its relevance to adults is questionable (Dietary Reference Intakes, Australian Ministry of Health). There have been animal studies showing that the ratio made no difference (Lueker & Lofgreen, Cramer). But in general it appears that a severe imbalance between calcium and phosphorus can be problematic, and it's wise to pay some attention to the balance of these nutrients. It is generally accepted in the reptile-keeping community that calcium supplementation is required to prevent calcium deficiency when feeding a diet of high-phosphorus insects (LLLReptile, DrsFoster&Smith).
Copyright 2015-2018 Carolyn Tielfan all rights reserved
